The Institute for Basic Science (IBS) research team led by Professor Kim Kimoon (CHEM, director of the Center for Self-assembly and Complexity at IBS) successfully visualized and controlled chemical reactions in a solution using audible sound for the first time.
Audible sound cannot directly affect chemical processes like bond formation or dissociation due to its low energy. Therefore, previous teams focused solely on the effect of sound on water surface movement or used sound to control floating and sediment particles. However, the IBS team hypothesized that if they could regulate gas solubility by controlling the pattern of sound vibrations, then they could form different chemical environments within the solution.
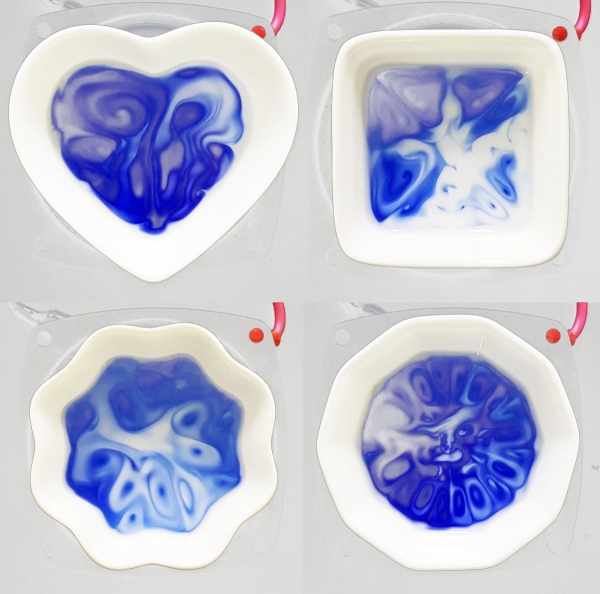
To visualize this, the team put various dishes above a speaker. First, they observed that due to small transverse vibrations, water waves in a Petri dish formed concentric circles. The wave patterns depended on the frequency and amplitude of the source sound and the geometry of the dish.
The team then poured O2-sensitive blue-colored methyl viologen radical cation into the dish. When a narrow range of bass sounds from 25-90 Hz was played, concentric patterns were formed. The nonmoving nodes remained blue, while the oscillating antinodes turned colorless. This was because sound vibrations continuously supplied energy to the antinodes, increasing active contact with oxygen at the air-water interface.
The experiment was repeated using the CO2-sensitive pH indicator, bromothymol blue (BTB). The researchers exposed the solution to carbon dioxide, which becomes acidic in water. Sound affected its solubility, so swirls of blue (basic), green (neutral), and yellow (acidic) partitions coexisted in the solution.
These patterns were self-healing—they became temporarily chaotic when disturbed, but soon returned to their original shape. Research Fellow Hwang Ilha commented, “We showed that the chemical environment of a solution can be segmented by controlling redox or acid-base reactions using sound, without a physical barrier. This can be useful in the development of smart pH-responsive chemical network systems within predetermined microenvironments, mimicking those operating inside our cells.” Another Research Fellow Rahul Dev Mukhopadhyay, invoked the tale of The Pied Piper, who used sound from his pipe to control rats. He said, “Even synthetic molecules can exhibit life-like behavior—listening and following a musical track.”
Prof. Kim added, “In the near future, we may further expand the use of audible sound from chemistry to other fields, such as physics, fluid mechanics, chemical engineering, and biology.”
The study was published in the journal Nature Chemistry on Aug. 11. Furthermore, the images taken during this study were presented at the fifth IBS Art in Science exhibition as Painting with a Sound Brush.


